SaferLung
- About
- Our solutions for you
- Our services
- Partner contribution
- Regulatory Framework
- Get further information
About
Lung health can be impaired by various compounds such as chemo therapeutics, smoke fume ingredients, fibers as known from asbestos as well as by radiation. This impairment can lead to acute or permanent toxicity through inflammation and fibrosis. Both inflammation and fibrosis lead to a decreased absorbance of oxygen in the lungs resulting in breathlessness during exercise, fatigue, dry cough, shortness of breath and discomfort or worsening symptoms when lying on the back [1].
Although severe lung damage in most cases cannot be reversed, there are several possibilities for treatment of lung toxicity symptoms such as corticosteroid-treatment to decrease inflammation and pain, oxygen-treatment to supplement oxygen, narcotics-treatment against pain and shortness of breath and approaches for rehabilitation to learn new breathing techniques for instance [1]. Nevertheless, such treatments are expensive and exhausting. Therefore, the best way to avoid lung toxicity is prevention and to avoid the exposure to toxins that harm the lung in advance to ensure lung health throughout lifespan.
The difficulty of characterizing lung toxins lies in the differences between rodent models and human lung function. Substances that are toxic in humans might be non toxic in rodents. Furthermore, the human lung consists of many different cell types that build a complex structure needed for gas exchange that is challenging to simulate ex vivo. [2]
To be able to identify toxins, models have been developed to face those challenges. Current models include 2D cell cultures, lung spheroids and organoids, decellularized ECM and Scaffold Based models, iPSC approaches as well as lung on a chip models. [2,3] As illustrated in Figure 1, such models cannot only be used to predict lung toxicity of compounds but also allow to personalise treatment after the lung has already been damaged.
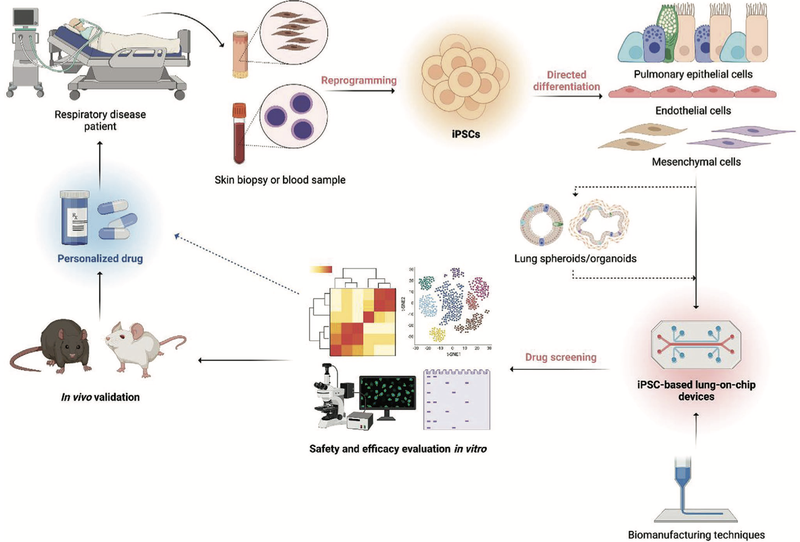
Figure1: Schematics of the cyclic process used for the application of lung-on-chip devices in personalized medicine. [2]
To be able to identify toxins before they can damage the lung, already existing data can be evaluated in in silico machine learning approaches. Based on feature extraction and ontology-based characterisation and testing, chemicals can be assessed for their toxic effect in humans.
At SaferWorldbyDesign we have developed a solution package specific for concerns of lung toxicity. Thereby we not only offer newest in vitro approaches, including multi organ on a chip technology, but also expertise in in silico modeling, machine learning, data management and knowledge infrastructure with which we build a framework for effective and efficient lung toxicity risk assessment.
References
[1] Hoafredericksburg, 28th March 2023 (Link)
[2] Advanced In Vitro Lung Models for Drug and Toxicity Screening: The Promising Role of Induced Pluripotent Stem Cells, by Anabela Moreira, Michelle Müller, Pedro F. Costa,* and Yvonne Kohl, 2022.
[3] A multiplex inhalation platform to model in situ like aerosol delivery in a breathing lung-on-chip by Arunima Sengupata, Mohammad Jamshidi, Magali Schwob, Widad Hassan, Lea Lara De maddalena, Andrea Hugi, Andreas O. Stucki, Patrick Dorn, Thomas M. Marti, Oliver Wisser, Janick D. Stucki, Tobias Krebs, Nina Hobi and Oliver T. Guenant in Frontiers in Pharmacology, 2023.
Our services
With our solutions we aim to help you in characterizing compounds in concern of potential harmful effects to the lung. We offer a variety of characterization approaches to characterize your compound under OECD or emerging guidelines in a tiered strategy. With this you have the possibility to design your products in a safe and sustainable way with regards to inhalation exposure.
In a first tier, we provide you with in silico approaches based on the following concepts:
In silico modelling: With our in silico service we develop algorithms for feature extraction and ontology characterization of new compounds based on already existing data from validated datasets under OECD guidance.
AI/Machine learning: We provide expertise for QSAR-based risk assessment of compounds which we can implement in a user friendly tool that is easy to handle.
Data Management: Data that adheres to FAIR principles and according to authority guidelines is key for data integrity and to be able to trust experimental outcomes. With our management systems, we provide harmonized and complete data. Workflows are organized, reproducible and accessible for future reference and securely managed through a premium cloud solution.
In a second or third tier approach we offer a variety of in vitro solutions specific for your needs and interests, such as Multi Organ on a Chip assays as provided by TissUse.
Partner contribution
In a second or third tier approach we offer newest in vitro technologies in collaboration with our partners. Services include:
TissUse
TissUse is a German based company which has developed a multi organ on a chip technique to replace animal experiments. Those chips can be used during drug development, in cosmetics, food, nutrition and consumer products to predict toxicity, ADME profiles and in vitro efficacy for example.
->Read more
Edelweiss Connect
Edelweiss Connect, as provider of the SaferWorldbyDesign platform, leverages its data expertise to assist in the modeling of toxicity. Using machine learning techniques Edelweiss Connect can help identify key markers and patterns that may show a substance’s potential to cause lung toxicity, allowing for better-informed decision-making in the drug development and risk assessment process.
> Read more
To guarantee the reliability of our characterization and assessment models, scientific reproducibility and data integrity have highest priority in our solution implementation. This approach not only underlies our methodology aligned with the current guidelines on lung safety assessment, but also takes into account what emerging regulatory guidelines will include based on new scientific progress.
- OECD guideline on inhalation testing
- European REACH framework