Human Induced Pluripotent Stem Cells
Major international initiatives have started to convert the traditional animal-based neurotoxicity tests to in vitro assays using both mammalian brain cells and human cells to detect and predict chemical hazards. Consequently, in line with legislations, there is an increasing need to develop alternative testing methods which could handle large numbers of drugs or chemicals with affordable time and cost and with human-relevant neurotoxicology (NT) outcomes. Development of new approach methods (NAMs), is important both for NT and developmental neurotoxicology (DNT) tests, providing data on the effect of chemicals and the potential adverse outcomes (AOs).
BioTalentum Ltd offers human induced pluripotent stem cell (hiPSC)-derived toxicology test service documented according to the ECVAM DB-ALM system (DB-ALM Protocol No. 207, 208 and 209) for screening the toxic effect of compounds. Beside the standard 2D assays, we also offer 3D neurosphere-based assays where the brain complexity is represented. The test system is available in a less differentiated (21 day-old) or a more differentiated (42 day-old) stage, where more mature neural cells with spontaneous synaptical activity can be investigated.
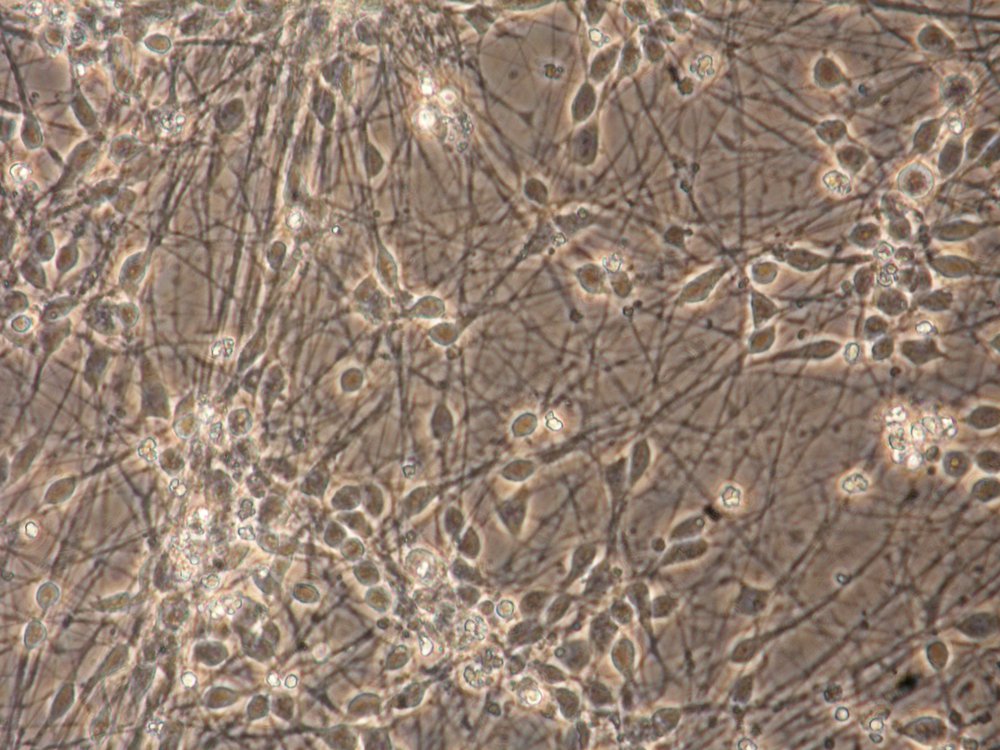
Mature hiPSC-derived neuronal-astrocyte-oligodendrocyte culture before toxicant treatment. Magnification: 20x
Solution benefits
- Replaces animal tests and provides more human-relevant results
- hiPSC-based neurons replace primary human brain cell samples
- The effect of compounds on different differentiation stages of the neuronal tissue can be investigated
- 3D Spheroids represent better the complexity of the neuronal tissue
Our service include:
- Consultation with customers, surveying their needs (no. of tested compounds, no. of dilutions, 2D/3D assay required, differentiation stage of neurons)
- hiPSC-derived neuron differentiation on a 96-well plate format according to the customer’s requirements. In general, 21 or 42 days of differentiated neural cultures are used, but the differentiation time can be altered upon request
- Compound treatment, no. of dilution, no. of biological replicates based on the negotiations with the customer. Paraquat, a well known neurotoxic agent is used as a positive control
- Incubation with the compounds for 24, 48 and 72 hours to test acute and 96 hours to test chronic exposure
- Viability assay (DB-ALM Protocol No. 207, 208, 209)
- Data analysis to determine the EC50 and IC10 values
- A report for the customer
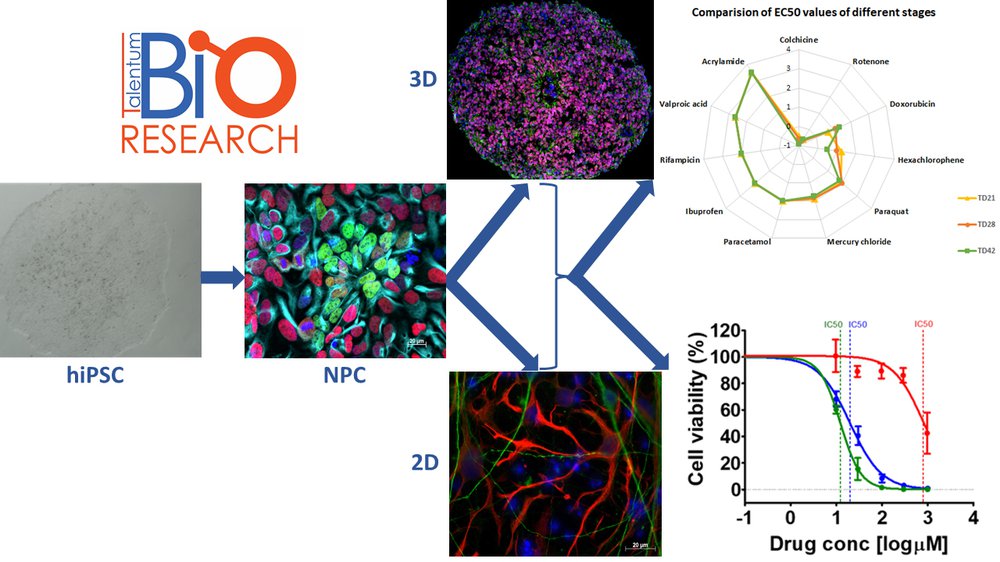
BioTalentum - workflow
Kobolak J., et al. Human induced pluripotent stem cell-derived 3D neurospheres are suitable for neurotoxicity screening. Cells. 2020 doi: 10.3390/cells9051122
Krebs A., et al. The EU-ToxRisk method documentation, data processing and chemical testing pipeline for the regulatory use of new approach method. Arch. Toxicol. 2020. doi: 10.1007/s00204-020-02802-6