Nonalcoholic fatty liver disease
An overview of NAFLD
Nonalcoholic fatty liver disease (NAFLD), the most common cause of chronic liver disease worldwide, is rapidly becoming the predominant cause of end stage liver disease and a leading indication for liver transplantation.
The prevalence rate of NAFLD is estimated at 25% in the general population with a significant increase expected in the coming decades.
The clinical syndrome of NAFLD spans from simple steatosis to severe life-threatening forms: nonalcoholic steatohepatitis (NASH), fibrosis, cirrhosis and hepatocellular carcinoma.
NAFLD is mostly asymptomatic and difficult to diagnose with no approved therapy. One of the most important limitation of NAFLD-drug development is the lack of human NAFLD models. Currently, the available NAFLD models lack accuracy or are too complex for high scale drug screening. In addition to their restricted use, diet- and genetic-based animal models may be misleading due to interspecies differences and may not mimic the multifactorial etiology of the human NAFLD. Thus, in vitro models may represent a robust and a suitable tool to investigate the molecular mechanisms of this disease and to screen a large number of potential drugs.
The HepaRG utility in NAFLD investigation
HepaRG cell line has proven its utility and efficiency as in vitro human cellular model for the induction of liver steatosis. Alongside, several accurate methods for the evaluation, quantification and characterization of fat accumulation i.e. quantification of intracellular lipids, expression profiling of transcription factors and lipid metabolism -related genes as well as steatosis-associated cell toxicity parameters, have been described in HepaRG cell model. Moreover, HepaRG cells are widely used to identify steatosis molecular biomarkers and to investigate the mechanisms and pathways underlying drug-induced steatosis contributing therefore to the development of new treatment strategies as well as to the screening of anti-steatotic drugs.
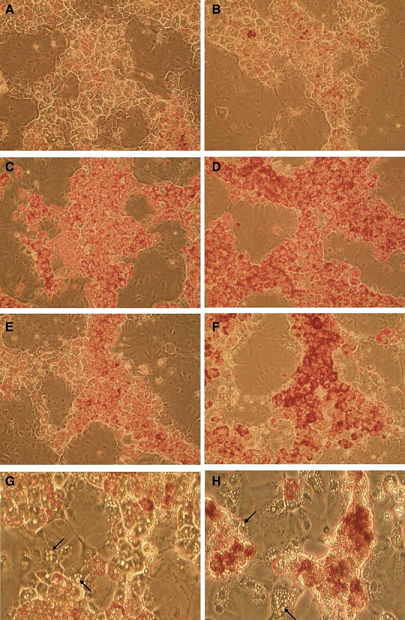
The first in vitro demonstration of drug-induced steatosis was performed in amiodarone and tertracycline-treated HepaRG cells (Figure 1, Antherieu, 2011). Detection of early cellular events (e.g., lipid accumulation, ROS generation, mitochondrial impairment) following short term exposure to steatogenic drugs is a major feature of HepaRG cell model. These events are evidenced at subcytotoxic concentrations of steatogenic drugs showing that they are primary specific effects to steatosis (Tolosa, 2016).
High sensitivity and specificity in the classification of steatogenic and non-steatogenic drugs were demonstrated in drug-exposed HepaRG cells (Lichtenstein, 2020, Allard, 2020, Tolosa, 2016).
Transcript and protein marker signatures for identification of steatogenic compounds were established in HepaRG cells and have shown a high concordance with in vivo findings (Lichtenstein, 2020).
Although the severe mitochondrial impairment has been identified as a leading cause of steatosis, other mechanisms such as the mild-to-moderate inhibition of mitochondrial fatty acid oxidation, the increased de novo lipogenesis and the impairment of very low-density lipoprotein have been shown to be involved in drug-induced steatosis in HepaRG cells (Allard, 2020). These findings help gain new insights into the steatosis mechanisms.
3D mono- and co-cultures of HepaRG cells as in vitro human liver model of NASH
There is growing evidence that 3D systems promote the maturity of hepatocyte phenotype which allows to better study the progression of a disease and increases the sensitivity of the system to detect hepatotoxic compounds.
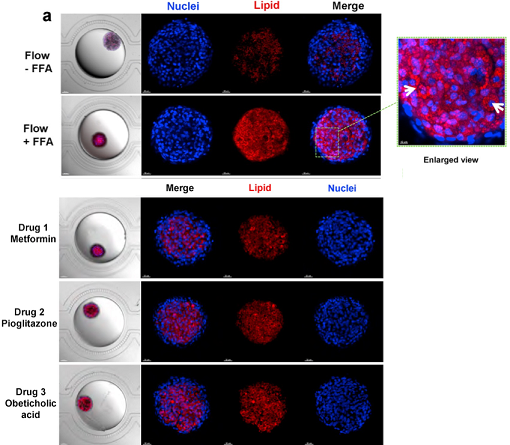
HepaRG organoids, differentiated in situ in a perfusion microfluidic device, SteatoChip, represent a robust model for recapitulating major NAFLD features (Teng, 2021). Under conditions mimicking the in vivo sinusoid architecture as well as the flow and 3D organoid culture, HepaRG organoids exhibit enhanced hepatic differentiation and functions as evidenced by albumin expression and secretion, urea synthesis, as well as by cell polarity and formation of bile canaliculi structures. Experimental applications such as organoids imaging for monitoring and analysis as well as sample collection for down-stream analysis such as qPCR and ELISA assays can be performed on HepaRG organoids.
In the context of steatosis modeling, a relatively high level of lipid accumulation was induced with free fatty acids in HepaRG organoids. Moreover, SteatoChip was used to assess the effect of three different anti-steatotic compounds. A reduction of 78.5%, 71.3% and 66.6% was noticed in metformin hydrochloride, pioglitazone hydrochloride and obeticholic acid treated HepaRG organoids respectively demonstrating that this chip could represent a scalable and sensitive drug testing platform as a response to the increasing need for anti-steatotic drug development and screening (Figure 2, Teng, 2021).
In parallel, liver multicellular systems represent interesting in vitro 3D cell models particularly when mimicking the hepatic lobule zonation of the in vivo liver.
HepaRG-based multicellular 3D hepatic steatosis model is a model mimicking the in vivo interplay between hepatocytes and non-parenchymal cells.
A pathologically relevant model using HepaRG cells, human umbilical vein endothelial cells (HUVECs), and mesenchymal stem cells (MSCs) was developed (Ryu, 2019). This model recapitulates NAFLD phenotype i.e. an increased expression of gluconeogenesis, lipogenesis, ER stress, and insulin resistance markers and can serve as a potent in vitro multicellular model for steatosis as well as other hepatic diseases investigation.
Another useful HepaRG-based system to monitor steatosis is generated by co-culturing HepaRG cells with HUVECs and human Kupffer cells (HKCs) (Suumond, 2019). Interestingly, the levels of lipid accumulation and ROS expression are higher in HepaRG-based spheroids cultured with free fatty acids supplements than in HepG2-based spheroids. Similarly, higher levels of proinflammatory cytokines are secreted by HepaRG in comparison to HepG2-based spheroids. These findings indicate that HepaRG-based model may better mimic the inflammatory status than HepG2-based model.
By comparing the reversibility of both systems, HepaRG showed fully recovered levels of accumulated lipids while HepG2 spheroids did not fully recover. Hence, the HepaRG-based model may represent more in vivo like condition and constitutes an appropriate model to monitor NAFLD (Suumond, 2019).
Altogether, HepaRG cell model, cultured in 2D or 3D configurations, represents a valuable alternative to in vivo models and primary human hepatocytes for steatosis induction and investigation of the underlying molecular mechanisms.