3D InSight™ Human Liver Microtissues
3D InSight™ Human Liver Microtissues are prequalified, long-lived liver models designed for drug safety and efficacy testing, and the study of healthy and diseased liver function. Composed of the primary human liver cells necessary for core liver functions, these microtissues are delivered assay-ready, in a lab-automation-friendly plate format ideal for screening.
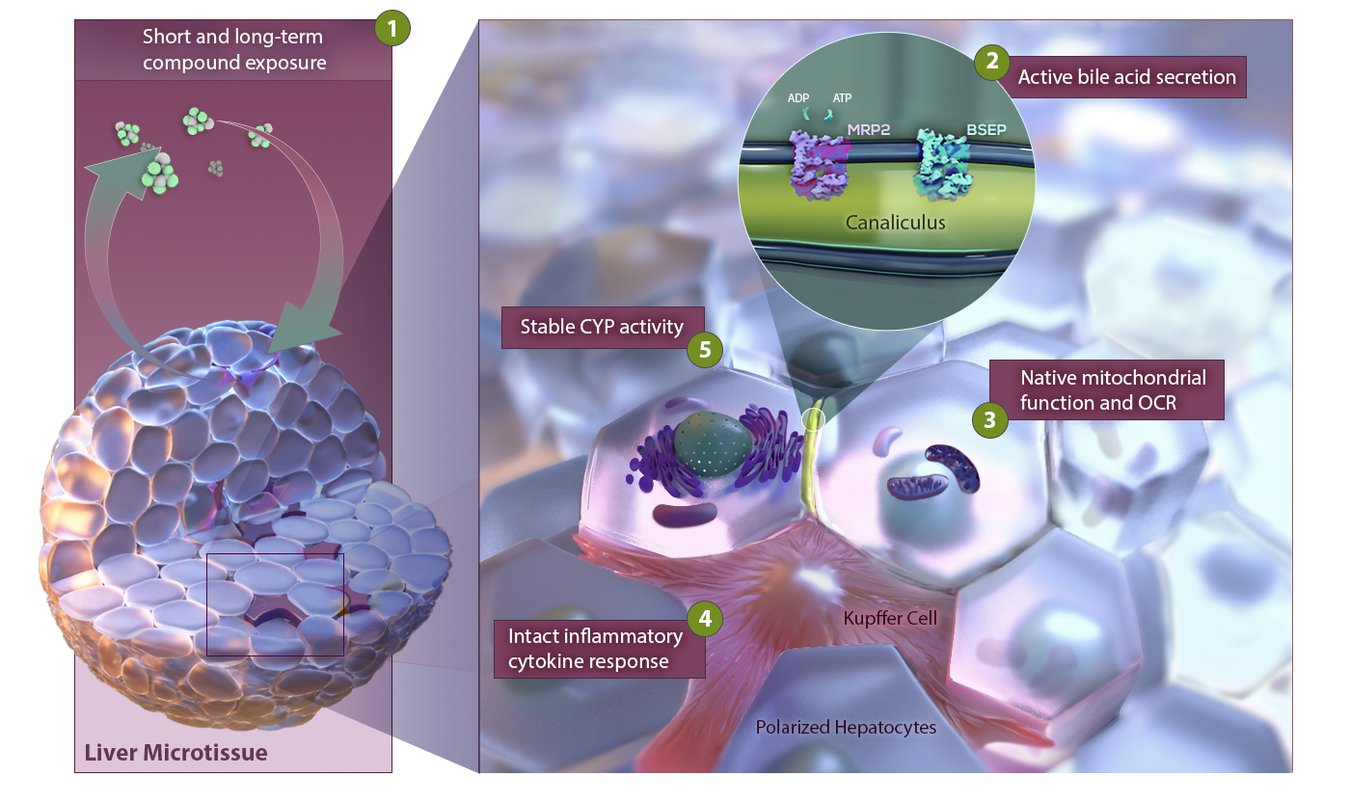
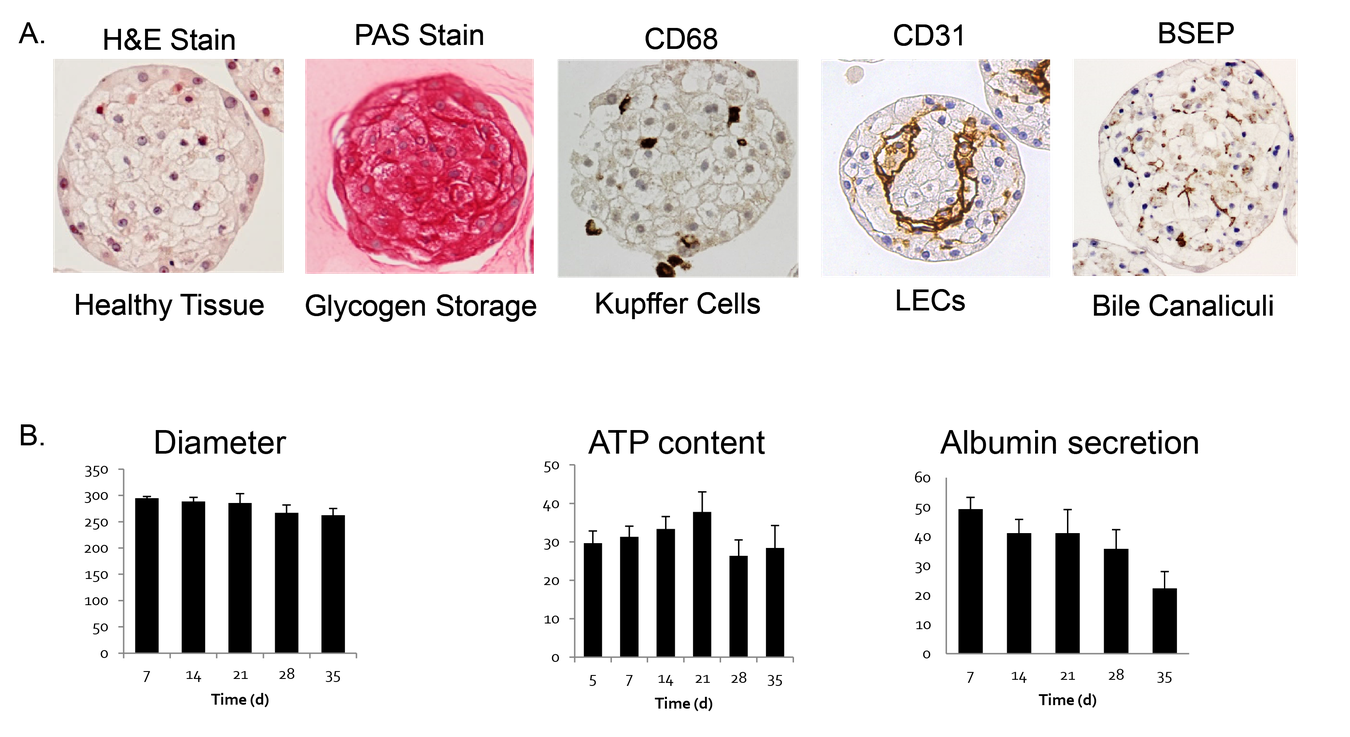
Pre-validated and standardized, 3D InSight™ Human Liver Microtissues have been thoroughly characterized on a functional, transcriptomic, and proteomic level. This model represents a functional and physiologically relevant in vitro liver model that maintains stable function for more than 5 weeks in culture and is therefore well suited for a broad range of liver research applications, such as DILI (drug induced liver injury) prediction.
InSight™ Human Liver Microtissues offer the most biologically- relevant, standardized, and scalable solution in the marketplace with proven performance and quality.
Classical 2D cell cultures have various limitations, as cultures are short lived (up to 3-5 days) not allowing long-term compound exposures and hepatocytes lose their organ specific characteristics due to un-physiological conditions. In contrast, 3D cultures are long lived (4 weeks and longer) and cells maintain organ-specific characteristics throughout the entire culturing time.
3D Solution Benefits
3D spheroid models bridge the gap from 2D in vitro models to the clinic providing more clinically relevant results.
A joint study (Proctor et al., 2017) conducted by collaborators at AstraZeneca and Genentech, resulted in a systematic validation of InSphero’s 3D InSightTM Human Liver Microtissues versus 2D primary human hepatocytes using a panel of 110 clinically known drugs. InSightTM Human Liver Microtissues showed a 2-fold higher sensitivity (61%) in identifying known hepatotoxicants compared to conventional 2D liver models.
The scalability of InSphero microtissues and easy handling of the AkuraTM plate technology allow for cost efficient processing and handling of 3D microtissues.
VISIT OUR SHOP
Order 3D InSight™ Human Liver Microtissues from InSphero
Messner et al. (2017) Applied In Vitro Toxicology, doi: 10.1089/aivt.2017.0022.
Proctor et al. (2017) Arch Toxicol, doi 10.1007/s00204-017-2002-1.